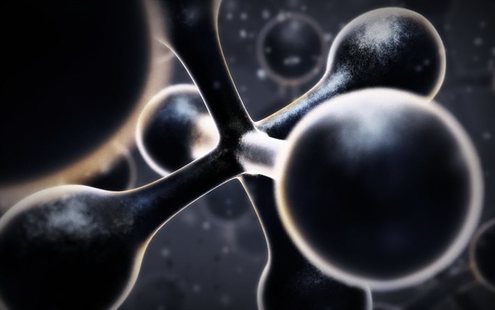
Molecules - are a group of atoms bonded together, representing the smallest fundamental unit of a chemical compound that can take part in a chemical reaction. ( pic below is an example of a molecule)
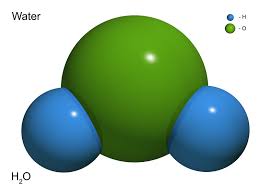
Compound- a thing that is composed of two or more separate elements; a mixture. ( below is a example of a compound.)
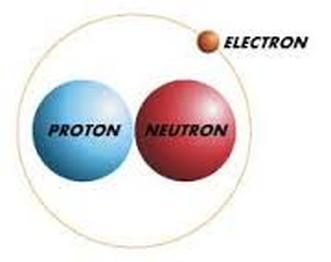
Protons - a stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron, but of opposite sign.
Electrons -a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
Neutrons - a subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen.
Electrons -a stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
Neutrons - a subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen.
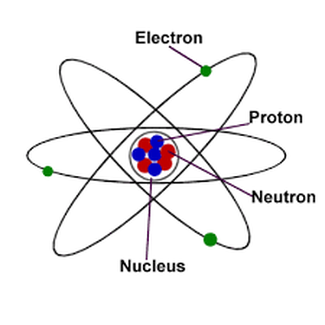
Atom is the smallest component of an element, characterized by a sharing of the chemical properties of the element and a nucleus with neutrons, protons and electrons.
Nucleus - the central part of an atom that contains the protons and neutrons.